In the realm of materials science, few minerals possess the structural complexity and versatile utility of zeolites. These hydrated aluminosilicates are defined by their microporous crystalline frameworks, an intricate network of channels and cavities that act as a host for water molecules and exchangeable cations. Among the more than forty naturally occurring zeolite species, clinoptilolite stands out for its abundance and exceptional physicochemical properties, making it a material of choice for applications ranging from radioactive waste remediation to agricultural soil amendment.
The defining characteristic of a zeolite's utility often lies in its interaction with water, specifically, the thermodynamics of hydration and dehydration. A recent study published in the journal Molecules has provided a sophisticated, quantitative analysis of these processes in clinoptilolite. By systematically exchanging the naturally occurring cations with potassium (K+), sodium (Na+), calcium (Ca2+), and magnesium (Mg2+), researchers have elucidated the profound influence of cationic species on the energy landscape of water sorption.
The Experimental Paradigm: Multi-Stage Thermal Analysis
To dissect the complex interplay between the zeolite framework, its extra-framework cations, and guest water molecules, the study employed a refined thermal analysis technique known as multi-stage Thermogravimetry and Differential Scanning Calorimetry (TG-DSC).
Standard thermal analysis typically involves a single, continuous heating ramp. While useful for determining total water content and thermal stability, this approach often obscures the nuanced, stepwise nature of dehydration. The multi-stage methodology used in this research subjected the cation-exchanged clinoptilolite samples to a programmed sequence of five heating stages, each followed by an isothermal holding period, up to a maximum temperature of 320°C. This was succeeded by a symmetrical five-stage cooling sequence.
This rigorous protocol allowed for the simultaneous measurement of mass changes (via TG) and heat flow (via DSC) under quasi-equilibrium conditions at specific temperature intervals. Crucially, it enabled the researchers to quantify not just the amount of water released or absorbed, but the specific enthalpy (heat energy) associated with these phase changes at different stages of hydration.
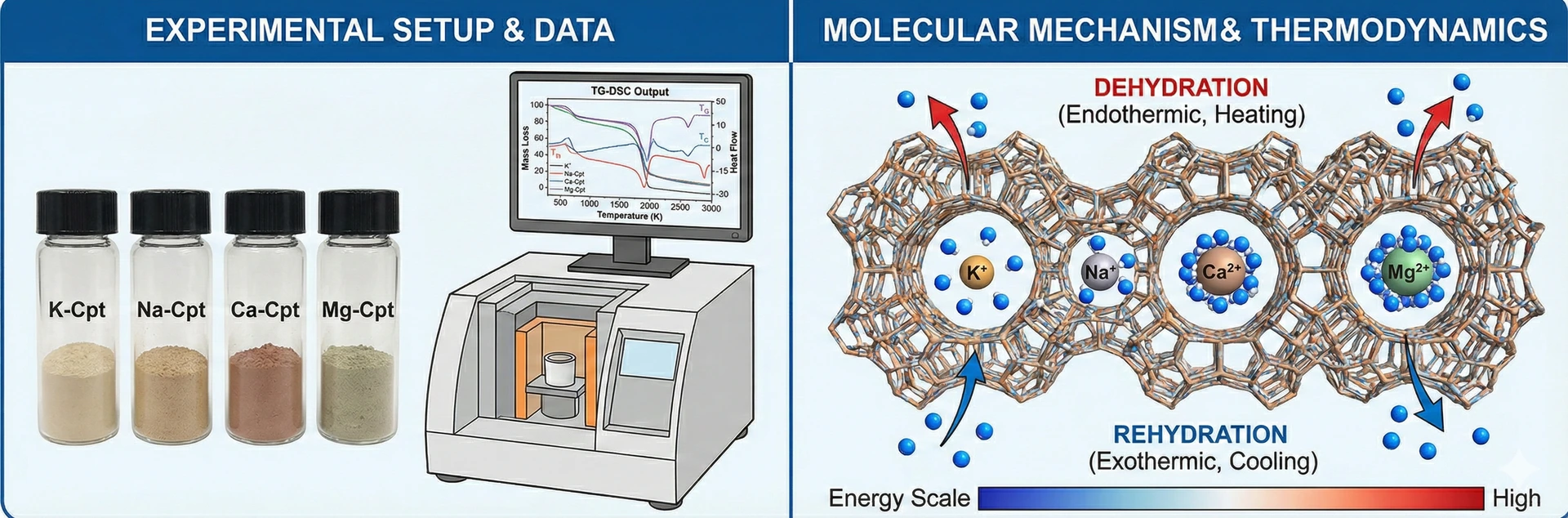
Cationic Charge and Dehydration Dynamics
The empirical data derived from the heating phase revealed a clear correlation between the valence of the exchangeable cation and the dehydration behavior of the clinoptilolite framework.
1. Monovalent vs. Divalent Cations: The study found a marked distinction in the total degree of dehydration achievable at 320°C. The samples exchanged with monovalent cations, K+ and Na+, exhibited a high degree of water loss, reaching approximately 90% and 94%, respectively. In contrast, the samples containing divalent cations, Ca2+ and Mg2+, were significantly more retentive, relinquishing only about 80% of their initial water content.
2. The Role of Charge Density: This phenomenon can be attributed to the fundamental principles of electrostatics. Divalent cations possess a higher charge density than their monovalent counterparts. Consequently, they exert a stronger electrostatic pull on the polar water molecules, forming more tightly bound hydration spheres. Organizations like the International Zeolite Association recognize this cation-water interaction as a primary determinant of a zeolite's adsorption properties. The energy required to overcome these strong attractive forces is substantially higher, resulting in incomplete dehydration under the experimental conditions.
3. The Enthalpy of Dehydration: Perhaps the most critical insight from the study was the quantification of the molar enthalpy of dehydration. The data showed that the energy required to remove a mole of water is not constant but increases incrementally as the zeolite becomes drier.
Initial Stages: The first water molecules to desorb are those occupying the central regions of the zeolite channels. These molecules are relatively loosely bound, primarily interacting through weaker hydrogen bonds with other water molecules and the framework oxygen atoms. Their removal requires less energy.
Final Stages: As dehydration progresses, the remaining water molecules are those directly coordinated to the extra-framework cations. Removing these molecules requires overcoming the significant electrostatic energy of the cation-dipole interaction.
This thermodynamic profile perfectly aligns with established crystallographic models that depict distinct populations of water molecules within the zeolite structure, ranging from mobile, bulk-like water to immobile, structurally bound water.
Novel Insights into Rehydration Thermodynamics
A unique and significant contribution of this research is the quantitative assessment of the rehydration process during the cooling phase. While dehydration is an endothermic process (requiring heat input), rehydration is exothermic (releasing heat).
By monitoring heat flow during controlled cooling, the researchers provided some of the first direct measurements of the enthalpy of hydration under dynamic conditions. The study revealed that the dehydrated clinoptilolite frameworks are highly reactive, rapidly readsorbing atmospheric moisture. The samples recovered approximately 60-66% of their original water content upon cooling back to ambient temperature. The measured heat release confirmed the energetic favorability of the hydration process and provided essential data for modeling the material's behavior in cyclic thermal applications.
Implications for Advanced Applications
The precise thermodynamic data yielded by this study has profound implications for the engineering of zeolite-based systems.
Thermal Energy Storage (TES): The reversible hydration/dehydration of zeolites forms the basis of thermochemical energy storage systems. Solar thermal energy can be used to dehydrate a zeolite bed during the day (storing energy), and the stored heat can be released upon rehydration at night. The study's detailed enthalpy values for specific cation forms are invaluable for designing highly efficient TES reactors, allowing engineers to select the optimal zeolite based on the desired operating temperature and energy storage density.
Industrial Desiccation and Gas Separation: For applications requiring ultra-low moisture levels, the findings point to the superiority of Na-exchanged clinoptilolite due to its ability to achieve a higher degree of dehydration. Conversely, for applications where strong water retention at higher temperatures is desirable, Ca- or Mg-forms may be preferable. This knowledge allows for the tailoring of desiccants for specific industrial processes, as noted in technical literature from sources like the U.S. Geological Survey.
Atmospheric Water Harvesting: The data on exothermic rehydration is directly relevant to the development of devices that harvest potable water from humid air. Understanding the energy kinetics of water adsorption is crucial for optimizing the cycle times and energy efficiency of such systems, particularly in arid regions.
Conclusion
The comparative multi-stage TG-DSC study serves as a powerful testament to the importance of fundamental thermodynamic research in materials science. By rigorously quantifying the energy dynamics of water interaction within cation-exchanged clinoptilolite, the research moves beyond qualitative descriptions to provide the hard data necessary for advanced engineering. It underscores that a zeolite is not a static entity but a dynamic system whose properties can be finely tuned through cationic manipulation. This level of understanding is essential for unlocking the full potential of these remarkable minerals in addressing contemporary challenges in energy, environment, and sustainability.
References and Further Reading:
Primary Research Article: Petrova, N.; Kirov, G.; Stanimirova, T. Comparative Multi-Stage TG-DSC Study of K+, Na+, Ca2+ and Mg2+-Exchanged Clinoptilolite Forms. Molecules 2025, 30, 4770. https://doi.org/10.3390/molecules30244770
International Zeolite Association (IZA): The premier global organization for zeolite science, providing extensive databases on zeolite structures, synthesis, and applications. https://www.iza-online.org/
U.S. Geological Survey (USGS) - Zeolites Statistics and Information: A definitive source for data on the occurrence, production, and industrial use of natural zeolites.
Auerbach, S. M.; Carrado, K. A.; Dutta, P. K. (Eds.). Handbook of Zeolite Science and Technology; CRC Press: Boca Raton, FL, USA, 2003. A comprehensive reference text covering the fundamentals of zeolite chemistry and engineering.
Cronstedt, A. F. "On an Unknown Mineral-Species Called Zeolites." (Original description of zeolites in 1756, highlighting their long history of scientific study).